What Is a Biological Indicator in Sterilization?
Release time: 2026-04-08
In the high-stakes environment of healthcare, infection prevention is not merely a protocol; it is a matter of life and death. According to the Centers for Disease Control and Prevention (CDC), healthcare-associated infections (HAIs) affect hundreds of thousands of patients globally each year. The frontline defense against these infections is the rigorous and flawless sterilization of surgical instruments and medical devices. But how can healthcare professionals be absolutely certain that a sterilization cycle was successful?
Enter the Biological Indicator (BI).
While chemical tapes may change color and mechanical gauges may show that proper temperatures were reached, only biological indicators provide definitive, physical proof that microbial life has been completely eradicated. This comprehensive guide will explore what biological indicators are, how they function in steam and Ethylene Oxide (EO) sterilization cycles, why they represent the gold standard per CDC and AAMI ST79 guidelines, and how global distributors can source high-quality, cost-effective BIs from top-tier manufacturers like Tianrun.
1. The Science Behind Biological Indicators
To understand what a biological indicator in sterilization is, we must look at the microbiological level. A biological indicator is a test system containing viable microorganisms providing a defined resistance to a specified sterilization process.
Unlike chemical indicators (which react to physical parameters like heat or steam) and mechanical indicators (the readouts on the sterilization machine itself), biological indicators test the actual lethality of the cycle.
1.1 The Role of Bacterial Endospores
BIs utilize bacterial endospores, which are dormant, highly resistant structures formed by certain bacteria to survive extreme environmental stress. Endospores are significantly harder to kill than the standard vegetative bacteria, viruses, or fungi found on used medical instruments. The logic is straightforward: if the sterilization cycle can kill a massive population of the most highly resistant spores known to science, it has undoubtedly killed all other less-resistant pathogens on the surgical instruments.
Most biological indicators contain a population of one million ($10^6$) spores. The specific type of spore used depends entirely on the sterilization modality being tested:
- Geobacillus stearothermophilus: Used for Steam and Hydrogen Peroxide (Plasma) sterilization. These spores are incredibly thermophilic (heat-loving) and offer extreme resistance to high-temperature steam.
- Bacillus atrophaeus: Used for Ethylene Oxide (EO) and Dry Heat sterilization. These spores are highly resistant to chemical alkylation and dry, arid environments.
2. Biological Indicators in Steam Sterilization Cycles
Steam sterilization, typically conducted in an autoclave, is the most common and dependable method used in hospitals and dental clinics globally. It uses saturated steam under pressure to achieve temperatures of 121°C (250°F) or 132°C to 135°C (270°F to 275°F).
2.1 How Steam BIs Work
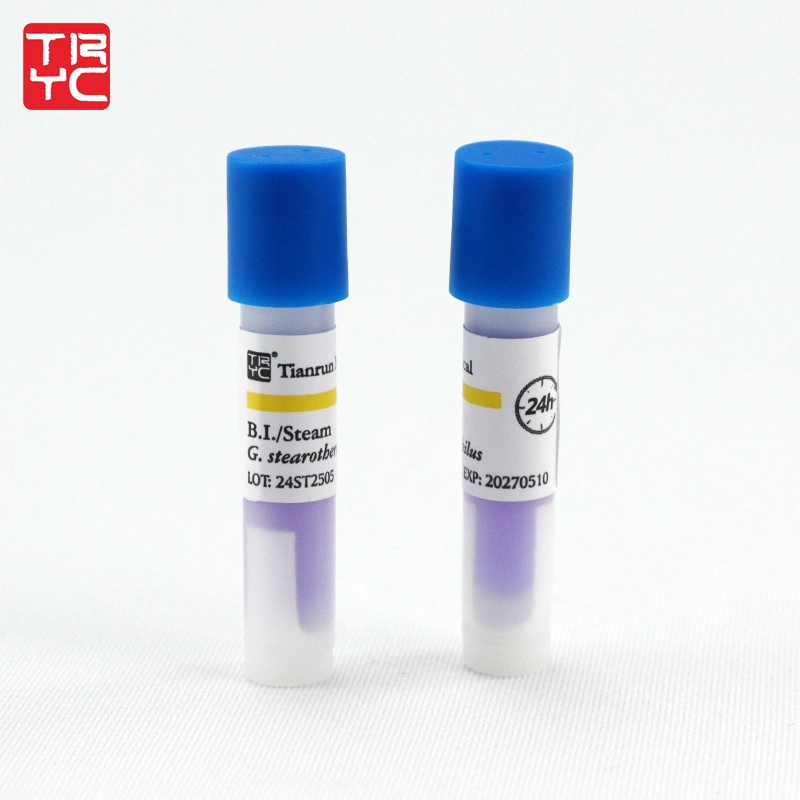
A steam biological indicator usually comes in the form of a Self-Contained Biological Indicator (SCBI). An SCBI consists of a plastic vial containing a spore strip of Geobacillus stearothermophilus and a small, crushable glass ampoule containing a growth medium (broth) with a pH indicator.
After the steam sterilization cycle is complete, the vial is removed. The operator crushes the inner glass ampoule, allowing the broth to mix with the spore strip, and places it in an incubator (usually set between 55°C to 60°C).
2.2 Interpreting the Results
- Negative Result (Pass): If the sterilization cycle was successful, all spores are dead. The liquid broth remains its original color (usually purple), and no fluorescence is detected in rapid-readout BIs. This confirms the load is sterile.
- Positive Result (Fail): If the cycle failed (due to air pockets, poor steam quality, or inadequate time/temperature), some spores survive. When incubated, these surviving spores consume the broth, producing acid. This lowers the pH, changing the liquid from purple to yellow, indicating a failed cycle.
3. Biological Indicators in Ethylene Oxide (EO) Sterilization
While steam is excellent for heat-resistant tools, many modern medical devices—such as flexible endoscopes, plastics, electronics, and custom procedure trays—are heat- and moisture-sensitive. Ethylene Oxide (EO) gas sterilization is the primary method for these items. EO kills microorganisms by alkylating their cellular DNA and RNA, preventing cellular reproduction.
3.1 How EO BIs Work
Because EO uses a chemical mechanism rather than extreme heat, the biological indicator uses Bacillus atrophaeus spores. Like steam BIs, EO BIs often utilize self-contained vials or paper spore strips wrapped in glassine envelopes.
3.2 The Unique Challenges of EO Verification
EO sterilization is highly complex, relying on four interdependent variables: gas concentration, temperature, relative humidity, and exposure time. A failure in any of these parameters can result in incomplete sterilization.
Because EO is used for highly complex, multi-lumen devices and large industrial pallets, the placement and verification of EO biological indicators are critical. The incubation temperature for Bacillus atrophaeus is typically lower than steam, around 37°C. While traditional EO BIs required up to 7 days of incubation, modern advancements have reduced this to 24-48 hours, or even 4 hours for rapid readout versions.
4. The Gold Standard: CDC and AAMI ST79 Guidelines
The use of biological indicators isn’t merely a recommendation; it is heavily regulated by global health authorities. In the United States, the CDC and the Association for the Advancement of Medical Instrumentation (AAMI) dictate the strict protocols for sterilization monitoring.
4.1 CDC Sterilization Guidelines
The CDC considers biological indicators the ultimate, “gold standard” proof of sterility assurance. According to CDC guidelines:
- Healthcare facilities must use BIs to monitor sterilizers at least weekly, though daily monitoring is strongly recommended for high-volume facilities.
- Crucial Rule for Implants: Every single load containing an implantable device (such as orthopedic screws, pacemakers, or artificial joints) must be monitored with a biological indicator, and the implant should ideally be quarantined until the BI results are confirmed negative.
4.2 AAMI ST79 Standards for Steam Sterilization
AAMI ST79 is the definitive comprehensive guide to steam sterilization and sterility assurance in health care facilities. AAMI ST79 mandates the use of Process Challenge Devices (PCDs).
A PCD is a standardized test pack that creates a significant challenge to the sterilization process. The biological indicator is placed inside the PCD. This ensures that if the steam can successfully penetrate the difficult PCD and kill the BI spores, it has easily sterilized the rest of the instruments in the load.
Placement Guidelines per AAMI ST79:
- The BI PCD should be placed in the most challenging area of the sterilizer to sterilize.
- For a standard steam sterilizer, this cold spot is typically on the bottom shelf, near the door, and directly over the drain line.
5. Proper Procedure: Frequency, Placement, and Recall Management
To maximize patient safety, central sterile services departments (CSSD) must adhere to flawless operational protocols when using BIs.
5.1 Step-by-Step BI Workflow
- Preparation: Place the BI inside a designated Process Challenge Device (PCD).
- Placement: Position the PCD in the sterilizer’s “cold spot” (e.g., over the drain).
- Execution: Run the standard sterilization cycle.
- Incubation: Remove the BI, activate it (crush the ampoule), and place it in the appropriate incubator alongside a “Control BI”.
- Note on Controls: A positive control BI from the same lot number that has not been sterilized must be incubated daily. This proves the spores were viable to begin with and the incubator is working correctly.
- Documentation: Record the lot number, sterilizer number, load number, and results in the facility’s sterility log.
5.2 What Happens When a BI Fails? (The Recall Protocol)
If a biological indicator yields a positive result (changes to yellow or fluoresces), it constitutes a severe protocol breach. The facility must immediately initiate a recall procedure:
- The sterilizer must be immediately taken out of service.
- All items processed in that sterilizer since the last known negative BI must be recalled.
- Recalled items must be completely reprocessed (cleaned, repackaged, and re-sterilized).
- The sterilizer must undergo maintenance and pass three consecutive BI tests in empty chamber cycles before being returned to service.
6. Sourcing Reliable Biological Indicators: The Role of Manufacturers Like Tianrun
Because biological indicators are life-critical Class II medical devices (in many jurisdictions), the manufacturing process requires extreme precision. Spore populations, D-values (the time required to kill 90% of the spores), and Z-values must be meticulously calibrated.
When healthcare facilities, hospital groups, and global medical distributors look for reliable sterilization monitoring tools, balancing uncompromising quality with cost-effectiveness is paramount. This is where established, large-scale manufacturers like Anhui Tianrun Packaging Co., Ltd. (Tianrun) play a pivotal role in the global medical supply chain.

6.1 Why Tianrun Stands Out in Sterilization Monitoring
Established in 1999, Tianrun has grown into one of China’s most prominent and respected manufacturers of medical packaging materials and sterilization consumables. For distributors seeking a reliable partner for BIs, Tianrun offers distinct advantages backed by hard data and robust infrastructure:
- Massive Production Scale: Tianrun operates out of an expansive 40,000-square-meter facility. Crucially, their manufacturing takes place within 100,000-grade cleanrooms, ensuring the sterile integrity of their biological and chemical indicators.
- Comprehensive Product Portfolio: Tianrun manufactures both steam and Ethylene Oxide (EO) biological indicators, as well as the accompanying self-contained incubators and Process Challenge Devices (PCDs). This allows hospitals and distributors to source a complete, integrated sterilization verification system from a single, reliable vendor.
- Strict Regulatory Compliance: Producing biological indicators requires adherence to the highest global standards. Tianrun operates strictly under ISO 13485 quality management systems specifically designed for medical devices. Their biological indicators conform to ISO 11138 standards, guaranteeing accurate spore populations and predictable resistance profiles.
- Global Supply Chain Efficiency: By sourcing directly from a leading manufacturer like Tianrun, global medical distributors can bypass middlemen, achieving highly competitive pricing without sacrificing the clinical reliability demanded by AAMI ST79 and CDC guidelines.
To explore their full range of medical packaging and sterilization monitoring solutions, professionals can review Tianrun’s robust capabilities and history directly on their official portal: Tianrun About Us.
7. Conclusion
A biological indicator in sterilization is much more than a simple compliance check; it is the ultimate safeguard between a patient and a potentially life-threatening healthcare-associated infection. By utilizing the incredible resistance of bacterial endospores like Geobacillus stearothermophilus and Bacillus atrophaeus, BIs provide the definitive scientific proof that a sterilization cycle—whether steam or EO—has successfully achieved its lethal objective.
Adhering to AAMI ST79 and CDC guidelines regarding the frequency, placement, and incubation of BIs is non-negotiable for modern healthcare facilities. Furthermore, as global healthcare demands rise, partnering with highly certified, large-scale manufacturers like Tianrun ensures that hospitals and distributors have uninterrupted access to the highest quality sterilization assurance products available on the market today.
Frequently Asked Questions (FAQs)
Q1: Can I release a sterilization load before the biological indicator results are fully incubated?
Answer: Generally, standard instruments can be released based on passing chemical and mechanical indicators. However, there is a strict exception: Implantable devices must never be released before the BI results are known. According to CDC and AAMI guidelines, loads containing implants must be quarantined until the biological indicator yields a negative (pass) result, ensuring patient safety during invasive surgeries.
Q2: What is the main difference between a Class 6 Chemical Indicator and a Biological Indicator?
Answer: A Class 6 chemical indicator reacts to physical conditions (time, temperature, and steam presence) by changing color, proving that the parameters for sterilization were met in that specific area. A Biological Indicator contains millions of live, highly resistant bacterial spores. It provides the only direct, physical proof that the sterilization cycle was lethal enough to actually kill microorganisms. Chemical indicators offer speed, while biological indicators offer definitive scientific proof.
Q3: Why do we have to incubate a “Control” biological indicator that was never put in the sterilizer?
Answer: Incubating a positive control BI from the same lot number is a critical quality assurance step. Because the control BI was not sterilized, it should show a positive result (bacterial growth and color change). This proves two things: first, that the spores in that specific manufacturing lot were alive and viable to begin with; and second, that your incubator is functioning at the correct temperature to promote growth. If the control BI fails to grow, the results of the sterilized BIs are considered invalid.